  
The cookies is used to store the user consent for the cookies in the category "Necessary". This cookie is set by GDPR Cookie Consent plugin. The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. Also, as you go down the periodic chart, the elements get bigger so the outer electrons get further away from the nucleus. Everything in the middle is somewhere in between.  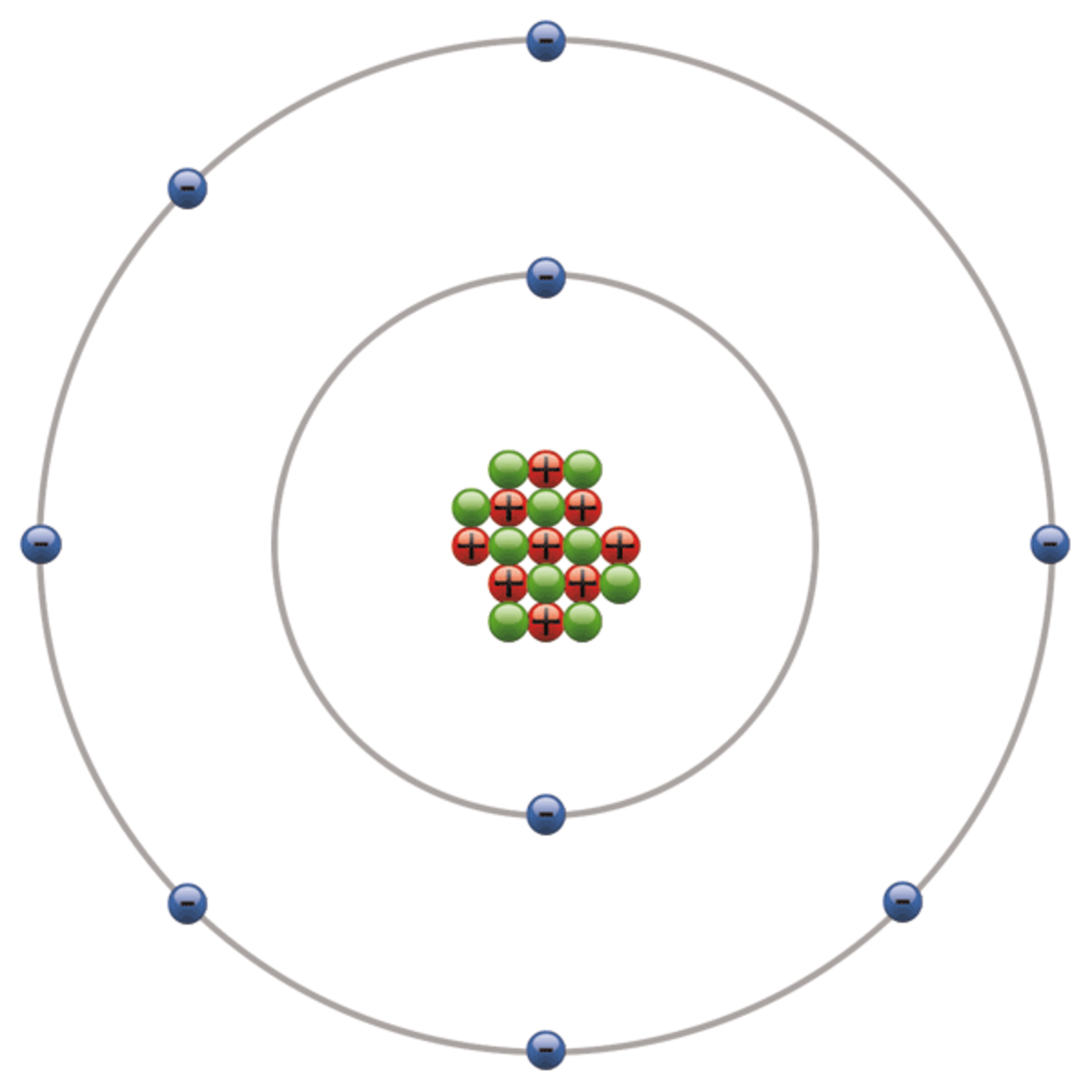
Non-reactive elements like those on the right of the periodic table have nice happy families of electrons that don’t need to make any new friends. How are reactive elements different from non reactive elements? So in general, the farther down and left you go, the more reactive the element will be. This means that they are even more “friendly” and they really want to react. Why are elements more reactive as they go down the periodic chart?Īlso, as you go down the periodic chart, the elements get bigger so the outer electrons get further away from the nucleus. For instance, fluorine (9) and chlorine (17) are short one electron from being the most stable and they are very reactive! The most reactive elements tend to be short one or two electron or have one or two extra. Which is the most reactive element in the universe? The two most reactive would be Fluorine and (theoretically) Francium. Those groups have 1 and 7 valence electrons, respectively, making them desperate to bond to something, to achieve a stable configuration of 8. The two most reactive groups of elements are the alkali metals and the halogens, because of their valence electrons. What are the two most reactive groups in the periodic table? Helium is a noble gas at the top of the noble gas family, which is group 18. Which is the least reactive nonmetal family? Reactivity of Group 1 Elements The reactivity of alkali metals increases from the top to the bottom of the group, so lithium (Li) is the least reactive alkali metal and francium (Fr) is the most reactive. What are the most reactive elements in order? The most reactive metals in the periodic tables are the alkali metals, followed by the alkaline earth metals. These groups easily give up their valence electrons to make a compound. Elements in Group 2 generally lose two electrons so their outer energy level is empty. Elements in Group 1 generally lose an electron so their outer energy level is empty. The most reactive metals are the elements in Groups 1 and 2. Where are the most reactive groups on the periodic table? The two most reactive families of elements are the halogens and the alkali metals. What 2 groups of elements are most reactive? In fact, they are the most reactive metals and, along with the elements in group 17, are the most reactive of all elements. Reactivity of Group 1 Elements Hydrogen is a very reactive gas, and the alkali metals are even more reactive. Which group of elements is most reactive? 5 Why are elements more reactive as they go down the periodic chart?.4 What are the two most reactive groups in the periodic table?.3 What are the most reactive elements in order?.2 Where are the most reactive groups on the periodic table?.1 Which group of elements is most reactive?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
